States of matter
Explore states of matter further with this helpful resource.
In the study of chemistry, matter is the essential substance that makes up everything in our universe. Learn the basics of matter using this resource, including its states, properties and classification.
Matter exists in three physical states: solid, liquid and gas.

Solid: The particles in a solid are packed tightly with a regular pattern. They are held in place, giving solids a fixed shape and a fixed volume.

Liquid: In a liquid, particles are closely packed but lack a regular pattern, allowing them to slide past one another. This gives liquids an indefinite shape that conforms to their container, while maintaining a fixed volume.

Gas: The particles in a gas move quickly and are far apart from each other. This allows gas to take the shape of the container by filling the space. Gases have both an indefinite shape and volume.
States of matter images, by RMIT, licensed under CC BY-NC 4.0
In solids, the particles are tightly packed and arranged in a regular pattern. The particles are locked in place and have minimal movement.
In liquids, the particles are closely packed but lack a regular pattern. The particles are more free to move.
In gases, the particles move quickly and are far apart from each other. The particles are very free to move.
Properties of matter can be classified into two categories depending on whether it is determined by changing the chemical identity or composition of the substance.
If the property can be observed/measured without changing the chemical identity of the substance, it is a physical property. Examples of physical properties include:
If the property can only be observed or measured by altering the chemical identity of the substance, it is a chemical property. Examples of chemical properties include:
Matter can be classified in many ways. You can use the flowchart shown and answer a series of yes/no questions to classify matter.
Are properties and composition constant? Pure substance (a substance that has a uniform chemical composition throughout, e.g. water, sugar, table salt). Can the pure substance be broken into simpler substances? Mixture (A physical combination of two or more pure substances, e.g. salt–sand mixture, seawater, concrete.) Does the mixture have a uniform composition throughout?
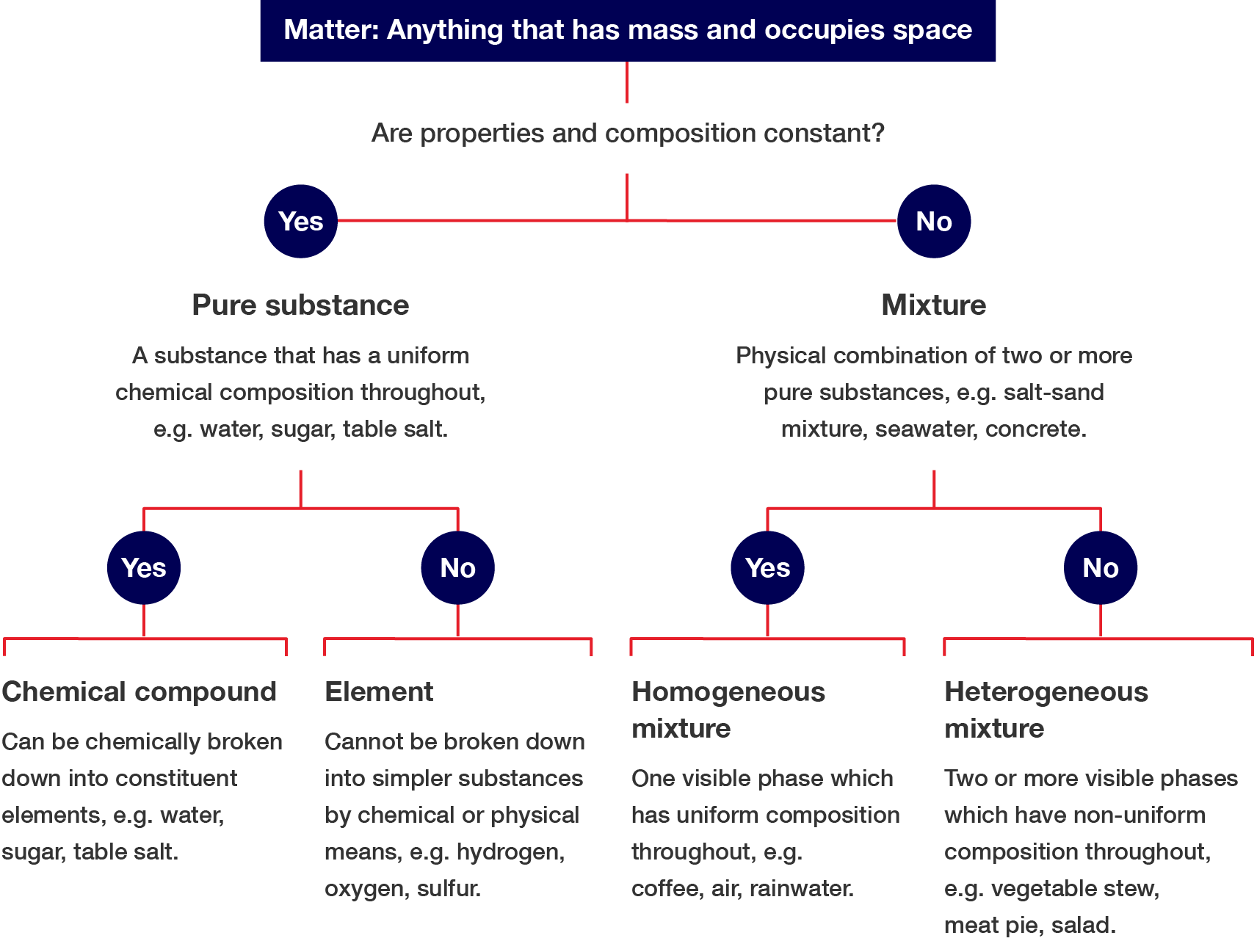
Matter: Anything that has mass and occupies space
Yes
No
Mixtures can be separated if we know the properties of their components, like state of matter.
In traditional times, the Gunditjmara Peoples of southwest Victoria used honeysuckle cones (banksias) as straws to filter water from muddy pools. The solid impurities would be trapped by the spikes, letting only clean drinking water through.

The Yindjibarndi Peoples of the Pilbara region in Western Australia used their understanding of density (how much mass is packed into a given volume) to separate large, less dense particles from smaller and denser ones. This process is called yandying. It involves placing a mixture in a wooden container and gently shaking it on an angle so that the smaller particles would fall out first, like gold panning. Yandying was commonly used to separate sand, dirt and ash from seeds.
Test yourself on your understanding of the states, properties and classification of matter.